 
Read more on this recall by clicking here. Inspired by simplicity and style, Qube breaks with tradition to. The Class II recall affects 742 total distributed devices-530 within the US and 212 internationally. Category: Medical Equipment & Devices Patient Monitoring Devices. In addition the monitor may fail to turn on due to an internal short. 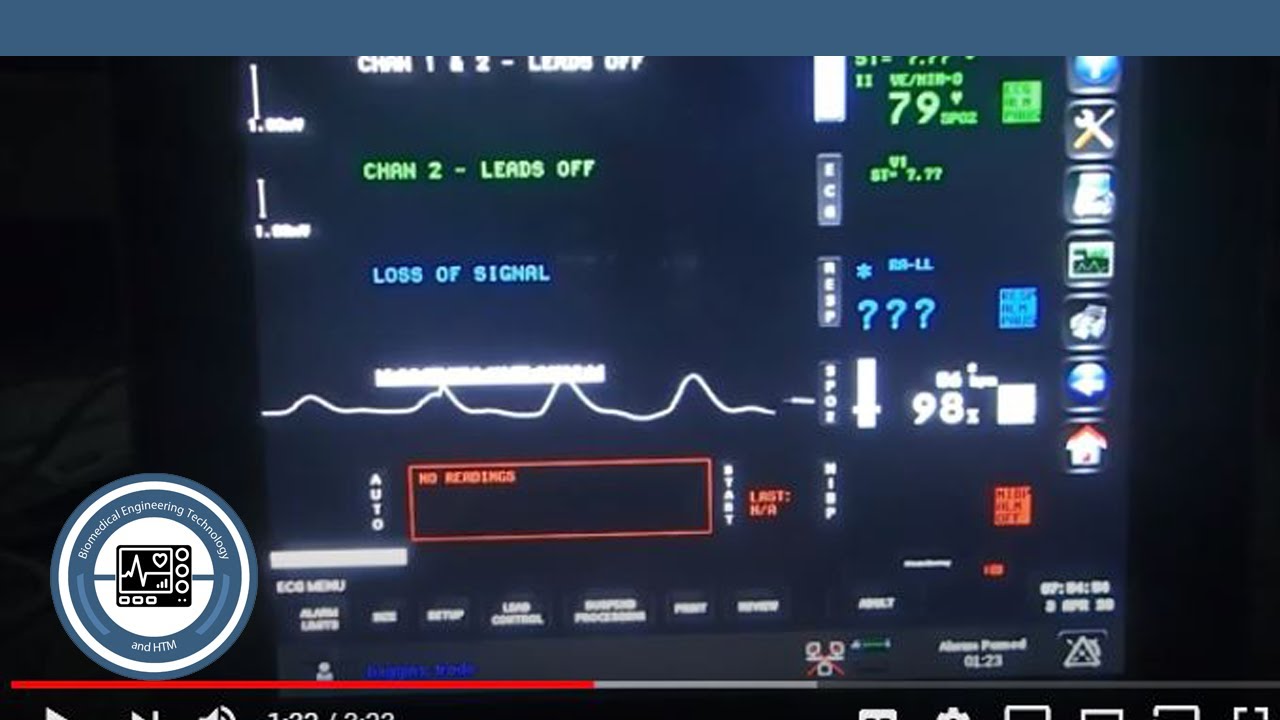
The monitor will function normally until the batteries become depleted and then the monitor will shut down. The company recalled the monitors when it learned through several reports that the input circuit may fail and the monitor will go on battery mode. The patient monitors are also capable of displaying alarm conditions on other monitors that are on the network through the alarm watch feature. compacto de cabecera Qube Spacelabs Healthcare WebUn vistazo rpido al monitor Qube. This Transport Monitor has a 12.1 inch LCD display with 1024 x 768 resolution and Dual Battery Slots. These devices determine a) when an alarm condition is violated b) the alarm priority (ie, high, medium, or low) c) alarm limits and d) when to initiate and terminate alarm notifications. WebEntdecke Spacelabs 91390 Qube Multiparameter Patientenmonitor. The Spacelabs Qube compact monitor 91390 Refurbished. Spacelabs Healthcare 91390 Qube Compact Bedside Monitor w/ 91496 Module. The Qube is intended to alert the user to alarm conditions that are reported by Spacelabs Healthcare parameter modules and/or other physiologic monitors via Flexport interfaces. The Spacelabs Healthcare Qube compact monitor 91390, functioning as either a bedside or central monitor, passively displays data generated by two Spacelabs Healthcare parameter modules, Flexports interfaces, and other SDLC-based products in the form of waveform and numeric displays, trends, and alarms. In the FDA Enforcement Report for the week of November 28, it lists the ongoing recall of the Qube compact monitor (patient monitor) 91390. 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
